 
Had been proposed, but none had been verified by experiment. Twentieth century, scientists were trying to answer this question. Or does an atom have some organized inner structure? At the beginning of the Particles randomly distributed inside the atom like blueberries in a muffin, How many of each a particular atom contains. So far, we have discussed electrons, protons, and neutrons and ways to determine TABLE 4.3 Relative abundance of naturally occurring isotopes of Table 4.3 lists the naturally occurring isotopes of several common elements, along with their relative abundance. Some elements have only one naturally occurring isotope. For example, 98.89% of all carbon occurring in nature is carbon-12, 1.11% is carbon-13, and only a trace is carbon-14. The isotopes of a given element are by no means equally abundant. We can write carbon-12, carbon-13, and carbon-14.Ībout 350 isotopes occur naturally on Earth, and another 1500 have been produced artificially. They can also be identified by the name of the element with the mass number of the particular isotope. The various isotopes of an element can be designated by using superscripts and subscripts to show the mass number and the atomic number. TABLE 4.2 The naturally occurring isotopes The composition of atoms of the naturally occurring isotopes of carbon are shown in Table 4.2. Isotopes are atoms that have the same atomic number (and are therefore of the same element) but different mass numbers. These different kinds of the same element are called isotopes. For example, some atoms of carbon (atomic number 6) have a mass number of 12, others have a mass number of 13, and still others have a mass number of 14. The atomic number and the mass number of an atom of an element can be shown by writing, in front of the symbol of the element, the mass number as a superscript and the atomic number as a subscript:įor example, an atom of gold (symbol Au), with an atomic number 79 and mass number of 196 is denoted as:Īlthough all atoms of a given element must have the same atomic number, they need not all have the same mass number. 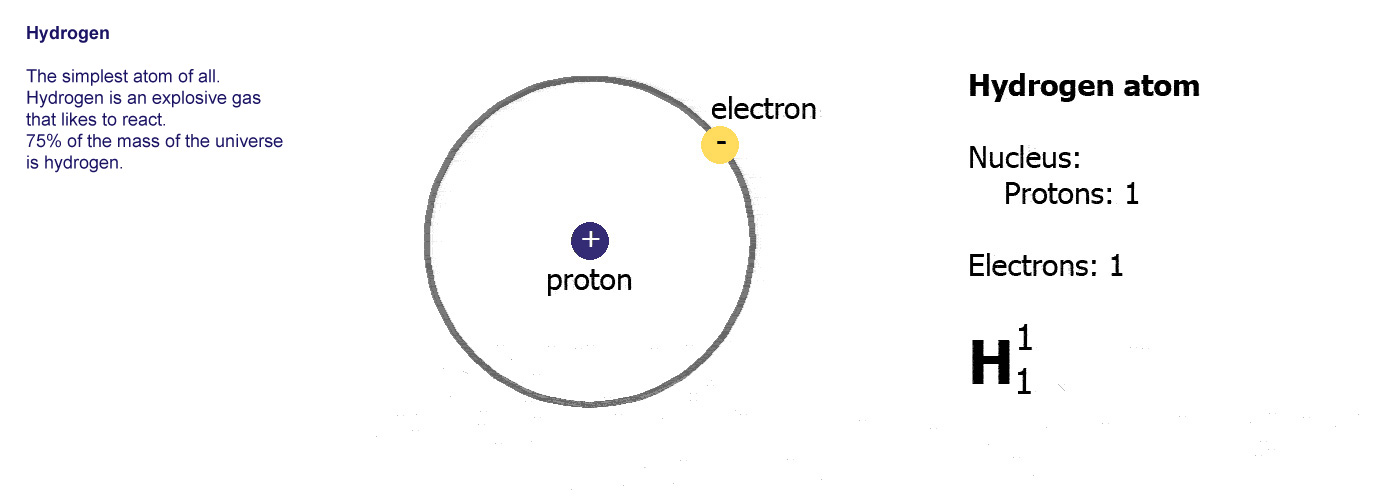
Mass number = number of protons + number of neutrons Number of neutrons = mass number - atomic number In other words, the number of neutrons in any atom is its mass number minus its atomic number. The mass number of an atom is equal to the number of protons plus the number of neutrons that it contains. Mass Number Equals Protons plus NeutronsĮach atom also has a mass number, denoted by the symbol A. The relationship between atomic number and the number of protons or electrons can be stated as follows:ī. Uranium has the atomic number 92 its atoms contain 92 protons and 92 electrons. Oxygen has the atomic number 8 its atoms contain 8 protons and 8 electrons. The atomic number has the symbol Z.ĭifferent elements have different atomic numbers therefore, atoms of different elements contain different numbers of protons (and electrons). It therefore also equals the number of protons in the nucleus and also equals numerically the number of electrons in the neutral atom. The atomic number equals the charge on the nucleus. The atomic numbers are listed along with the names and symbols of the elements on the inside cover of the text. Atomic Number Equals Electrons or Protons 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
